Bitter Rivals: Kratom’s Three-Ring Circus
/By Pat Anson
In recent years, you’ve probably come across stories about kratom, an herbal supplement used by millions of Americans for pain relief and to help manage conditions such as anxiety and depression.
While the vast majority of consumers use kratom safely, there is growing concern about outlier cases where kratom is abused or has even been associated with overdoses.
That has led to several states and dozens of cities and counties banning natural leaf kratom or a potent, concentrated kratom alkaloid called 7-hydroxymitragynine (7-OH). In some cases, they’re banning both.
Less well known is that the growing controversy over kratom is being fueled, in part, by an ongoing turf war between three rival industry-funded advocacy groups. Or, to use another cliche, a three-ring circus.
In one ring is the American Kratom Association (AKA), an organization of kratom manufacturers and vendors that sell natural leaf kratom products.
In another ring is the Holistic Alternative Recovery Trust (HART), which represents 7-OH manufacturers like American Shaman.
In the third ring is the Global Kratom Coalition (GKC), which was founded by JW Ross, who made a fortune selling a popular kratom-kava shot called “Feel Free.”
Like the AKA, the GKC favors natural leaf kratom, and takes a pugnacious approach to critics and competitors. The GKC likens rival 7-OH products to “powerful prescription opioids” that should be banned or heavily regulated.
You would think the AKA and GKC would be on the same team, since they both want to keep natural leaf kratom legal and accessible. But they are bitter rivals.
In an open letter, AKA chairman Matt Salmon claimed that GKC founder Ross is actually Jerry Cash, a convicted embezzler, who is trying to “make the AKA look bad” by misstating its position on kratom product formulations.
Salmon, a former congressman, also accused the GKC of launching a smear campaign against Mac Haddow, an AKA lobbyist who had his own run-ins with the law. Salmon’s letter is nearly three years old, but helps explain what is happening today.
“The most recent attacks against AKA and the personal attacks on Mac Haddow came after several ambush interviews orchestrated by Mr. Ross and his PR team providing incomplete, mischaracterized, and demonstrably false information to reporters,” said Salmon.
Kratom vs 7-OH
All three kratom groups accuse each other of jeopardizing what has become a lucrative business opportunity: selling an herbal supplement that is still largely unregulated by the federal government. Currently estimated to be worth over $2 billion, the global kratom industry is projected to grow to nearly $8 billion by 2032.
The latest example in this turf war is a self-styled “consumer alert” by the AKA warning about the “growing proliferation of dangerous products” containing 7-OH and other synthetic products made from kratom.
“Consumers deserve to know the truth,” said Haddow, Senior Fellow on Public Policy for the AKA, in a press release. “These 7-OH products are not traditional kratom. They are being engineered, concentrated, and marketed in ways that create risks that are not associated with natural kratom leaf.”
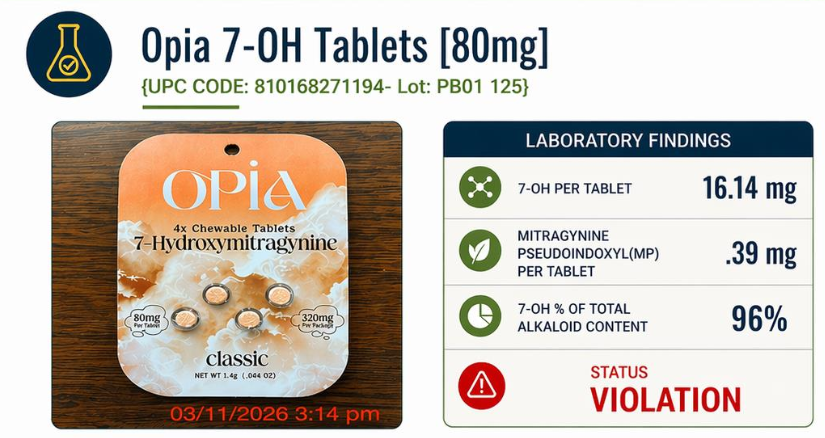
As evidence, the AKA cites lab results commissioned by the Texas Attorney General, which found that several 7-OH products contain alkaloid concentrations that exceed safety limits under state laws modeled after the Kratom Consumer Protection Act (KCPA) – legislation that the AKA has lobbied Texas and 20 other states to enact.
One such product, Opia 7-OH tablets, contain over 16mg of 7-OH – which is 96% of their total alkaloid content – well above the 2% limit under the KCPA.
AKA IMAGE
Critics of the KCPA say the law is misleading, doesn’t protect consumers, and is designed primarily to protect the financial interests of the AKA and other vendors who sell natural leaf kratom.
Not surprisingly, the AKA disagrees. It wants 7-OH scheduled as a controlled substance and a ban on 7-OH being marketed as “kratom” – moves that would preserve the legal status of natural leaf kratom.
“This is not a debate about kratom,” says Haddow. “This is about stopping a new class of unregulated, opioid-like substances from being disguised as something they are not.”
The Holistic Alternative Recovery Trust takes issue with the portrayal of 7-OH as a dangerous opioid.
“7-OH is not a synthetic substance or a novel additive, it is a naturally occurring alkaloid found in the kratom leaf itself. Calling it anything else misrepresents the science,” a HART spokesperson said in a statement to PNN.
“This is part of a broader pattern we’ve seen from the American Kratom Association and the Global Kratom Coalition, misrepresenting the science in ways that benefit certain segments of the market, particularly whole-leaf producers, while dismissing or sidelining millions of consumers who rely on 7-OH products.”
‘Kratom Is an Opioid’
The AKA’s latest attack on 7-OH comes on the heels of the GKC’s endorsement of a bill in Congress that would target “lab-made opioids” by amending the Controlled Substance Act to include 7-OH as a Schedule One substance, in the same category as heroin.
The association with opioids is a bit of a canard, but it makes for a good headline. Kratom comes from the leaves of the Mitragyna speciosa tree, which has more in common with coffee than it does with poppy plants, from which opioids such as heroin are produced.
Like kratom, coffee and other comfort foods such as chocolate stimulate endorphin nerve receptors and have mild “feel good” effects. You might even say they have “opioid-like” effects. But that doesn’t make Hershey’s Kisses opioids or Mrs. Olson a drug dealer.
Squabbling over the safety of each other’s products has not benefited kratom consumers and has contributed to sensational reporting about kratom and 7-OH causing addiction and overdoses.
The latest example appears in The Conversation, which commissioned Dr. Andrew Kolodny to write an op/ed about kratom. Kolodny is the founder and president of Physicians for Responsible Opioid Prescribing (PROP), an influential anti-opioid activist group.
Kolodny used a misleading CDC study about calls to poison control centers to paint a misleading portrait of kratom as just another opioid.
“For now, the evidence shows that kratom is an opioid with real risks – not a harmless supplement,” wrote Kolodny, while shamelessly ignoring his own role in restrictions on the use of opioid medication, which led to greater use of kratom. “Kratom’s rising use over the past decade coincided with the opioid crisis, as people searched for alternatives to prescription opioids.
“Some in the kratom industry argue that only newer products with boosted levels of 7OH are dangerous. But the evidence does not support that claim. Deaths linked to kratom were already rising before the newer 7OH products appeared on the market in late 2023.”
The three kratom advocacy groups – who all favor limited regulation – would be wise to consider that demonizing each other’s products only contributes to state after state and city after city enacting kratom bans.
“There are legitimate concerns in the marketplace, particularly around inconsistent products, unclear labeling, and lack of transparency. Those issues deserve attention. Consumers should know exactly what they are purchasing through clear labeling, verified third-party testing, and honest disclosure of potency and contents,” says HART.
“But the solution is not to single out or ban one compound based on flawed narratives. Policymakers should instead focus on enforceable manufacturing standards and practical safeguards, such as milligram-per-serving limits, standardized labeling, and quality controls, rather than arbitrary caps that risk eliminating products people currently rely on.”
Last summer, the FDA said it would ask the DEA to have 7-OH – but not whole leaf kratom – classified as an illegal Schedule One controlled substance.
The DEA, which doesn’t even mention kratom or 7-OH in its most recent National Drug Threat Assessment, has yet to move forward on the FDA’s request. Perhaps federal agencies are just as divided about kratom as kratom advocates are.